Dorfman AL, Cuenca N, Pinilla I, Chemtob S, Lachapelle P. Immunohistochemical evidence of synaptic retraction, cytoarchitectural remodeling, and cell death in the inner retina of the rat model of oygen-induced retinopathy (OIR). Invest Ophthalmol Vis Sci. 2011, 52(3):1693-708.
Scientific impact: Retinopathy of prematurity (ROP) is one of the main cause of vision loss in newborns and is induced by anarchic retinal neo-vascularization. It is a complex disease, and lack of knowledge prevents complete understanding of its development. This article proposes a new mechanism of cell death and synaptic retraction at the origin of the hyperpoxic damage in ROP. In addition, this work strongly suggests that genetic factors can cause a greater sensitivity to hypoxic stress amongst individuals. This novel mechanism will improve our understanding of ROP development and could lead to new therapeutic avenues.
Network contribution: This work was a collaboration between many members of the VHRN and international researchers (Cuenca N, Pinilla I, Espagne). They were partly financed by the VHRN (retinas from the ocular tissue bank and bursaries).
* * *
Original abstract
Purpose: Postnatal exposure to hyperoxia destroys the plexiform layers of the neonatal rat retina, resulting in significant electroretinographic anomalies. The purpose of this study was to identify the mechanisms at the origin of this loss. Methods: Sprague-Dawley (SD) and Long Evans (LE) rats were exposed to hyperoxia from birth to postnatal day (P) 6 or P14 and from P6 to P14, after which rats were euthanatized at P6, P14, or P60. Results: At P60, synaptophysin staining confirmed the lack of functional synaptic terminals in SD (outer plexiform layer [OPL]) and LE (OPL and inner plexiform layer [IPL]) rats. Uneven staining of ON-bipolar cell terminals with mGluR6 suggests that their loss could play a role in OPL thinning. Protein kinase C(PKC)-α and recoverin (rod and cone ON-bipolar cells, respectively) showed a lack of dendritic terminals in the OPL with disorganized axonal projections in the IPL. Although photoreceptor nuclei appeared intact, a decrease in bassoon staining (synaptic ribbon terminals) suggests limited communication to the inner retina. Findings were significantly more pronounced in LE rats. An increase in TUNEL-positive cells was observed in LE (inner nuclear layer [INL] and outer nuclear layer [ONL]) and SD (INL) rats after P0 to P14 exposure (425.3%, 102.2%, and 146.3% greater than control, respectively [P < 0.05]). Conclusions: Results suggest that cell death and synaptic retraction are at the root of OPL thinning. Increased TUNEL-positive cells in the INL confirm that cells die, at least in part, because of apoptosis. These findings propose a previously undescribed mechanism of cell death and synaptic retraction that are likely at the origin of the functional consequences of hyperoxia.
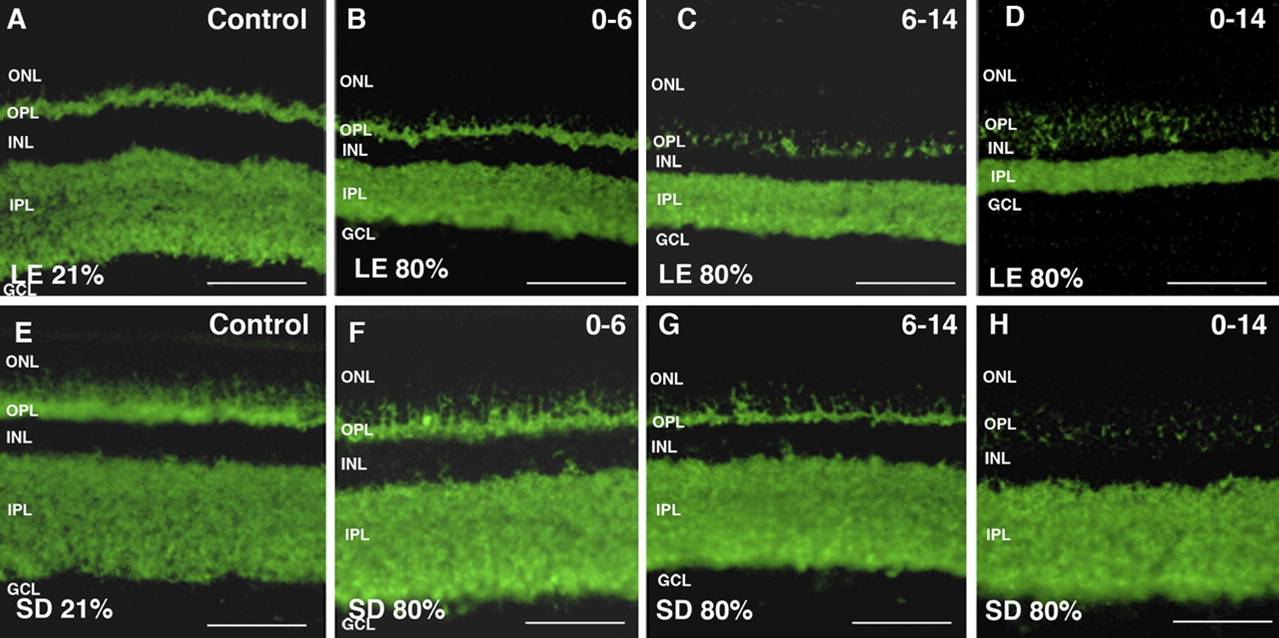
Immunofluorescent staining indicates a loss of neural cells in the retina of two species of rats (Long Evans A-D and Sprague Dawley E-H) during prolonged hyperoxia exposure