Dejda A, Mawambo G, Cerani A, Miloudi K, Shao Z, Daudelin JF, Boulet S, Oubaha M, Beaudoin F, Akla N, Henriques S, Menard C, Stahl A, Delisle JS, Rezende FA, Labrecque N, Sapieha P. Neuropilin-1 mediates myeloid cell chemoattraction and influences retinal neuroimmune crosstalk. J Clin Invest. 2014 Nov 3;124(11):4807-22.
Scientific impact :
Diabetic retinopathy is the most prominent complication of diabetes and the leading cause of blindness in working age individuals. It is estimated that 500 000 Canadians are afflicted by diabetic retinopathy and it is predicted that the incidence will double over the next 15 years.
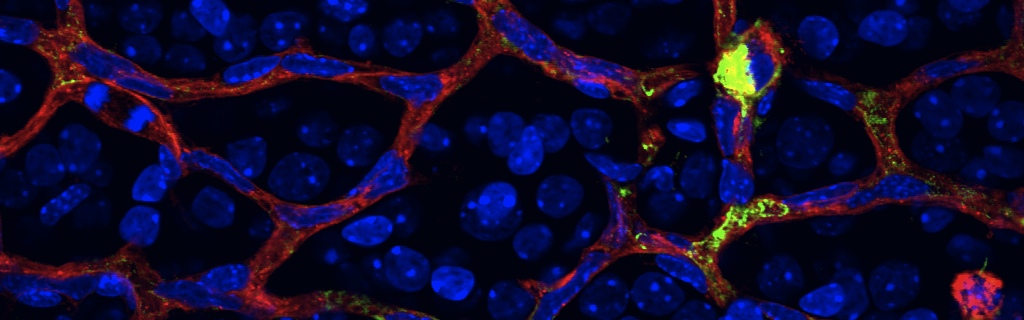
In our study, we describe a novel mechanism by which severely stressed neuronal cells in the diabetic retina provoke destructive inflammation in the eye. We demonstrate that via the multi-ligand receptor Neuropilin-1, Vascular Endothelial Growth Factor and Semaphorin3A act as potent attractants for a specialized population of immune cells (mononuclear phagocytes) that later promote the exaggerated pathological vessel growth associated with disease progression. Importantly, we provide evidence for a novel pharmacological intervention that reduces the inflammation associated with pathological retinal vessel growth. Our findings also suggest that current anti-VEGF therapies (a popular treatment for ocular vascular diseases) may in part be effective by reducing destructive ocular inflammation.
Network contribution: Granting of a recruitment award to Gaelle Mawambo (co-first author), a recruitment award to Augustin Cerani (third author) and an Initiation to vision student research award to Felix Beaudoin (ninth auteur).
* * *
Original abstract
Immunological activity in the CNS is largely dependent on an innate immune response and is heightened in diseases, such as diabetic retinopathy, multiple sclerosis, amyotrophic lateral sclerosis, and Alzheimer’s disease. The molecular dynamics governing immune cell recruitment to sites of injury and disease in the CNS during sterile inflammation remain poorly defined. Here, we identified a subset of mononuclear phagocytes (MPs) that responds to local chemotactic cues that are conserved among central neurons, vessels, and immune cells. Patients suffering from late-stage proliferative diabetic retinopathy (PDR) had elevated vitreous semaphorin 3A (SEMA3A). Using a murine model, we found that SEMA3A acts as a potent attractant for neuropilin-1–positive (NRP-1–positive) MPs. These proangiogenic MPs were selectively recruited to sites of pathological neovascularization in response to locally produced SEMA3A as well as VEGF. NRP-1–positive MPs were essential for disease progression, as NRP-1–deficient MPs failed to enter the retina in a murine model of oxygen-induced retinopathy (OIR), a proxy for PDR. OIR mice with NRP-1–deficient MPs exhibited decreased vascular degeneration and diminished pathological preretinal neovascularization. Intravitreal administration of a NRP-1–derived trap effectively mimicked the therapeutic benefits observed in mice lacking NRP-1–expressing MPs. Our findings indicate that NRP-1 is an obligate receptor for MP chemotaxis, bridging neural ischemia to an innate immune response in neovascular retinal disease.
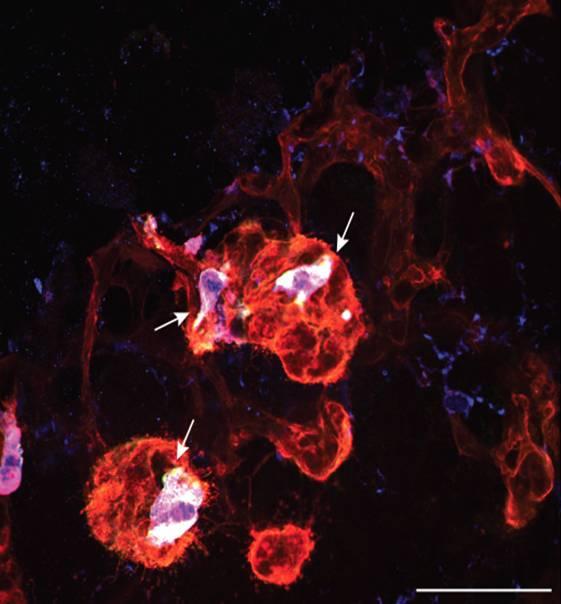
Pathological neovascular tuft (red) in a model of proliferative diabetic retinopathy with neuropilin-1-positive microglia (white)